-
 Welcome To Vacsera
Welcome To Vacsera -

-
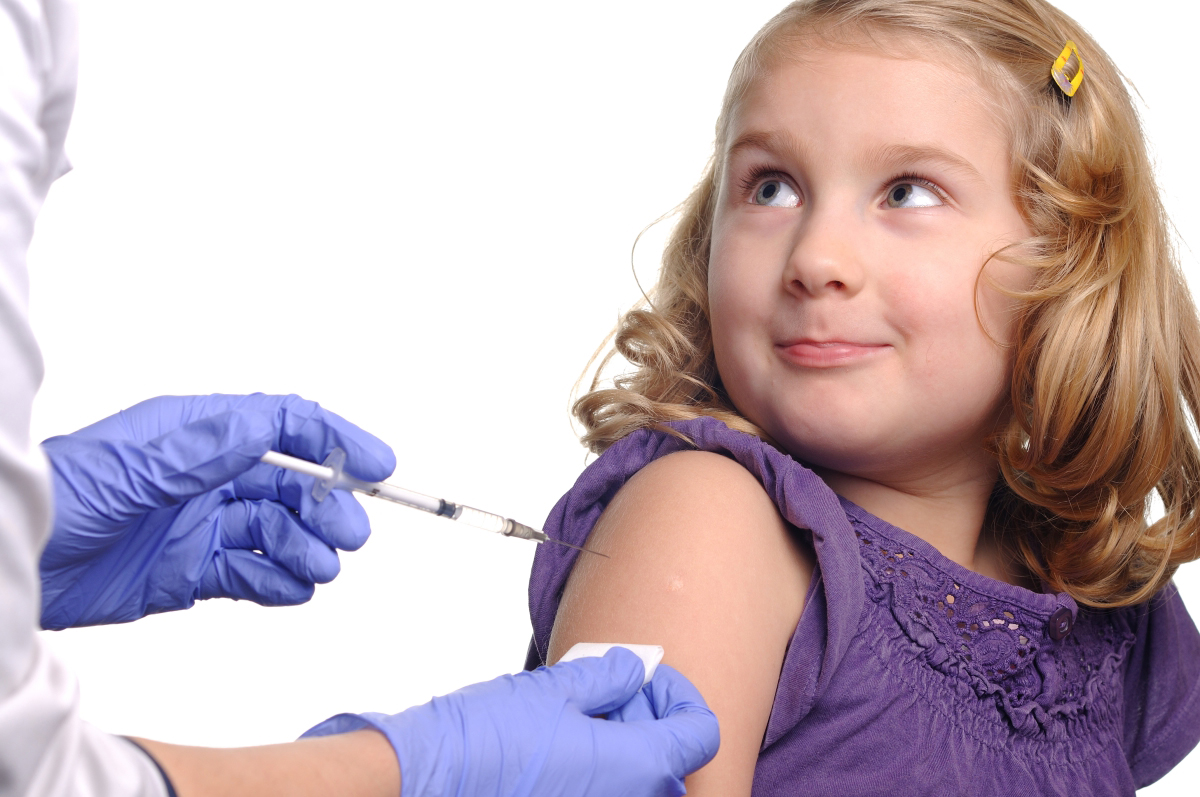 Vaccines Services
Vaccines Services -
 Blood Services& its derivatives
Blood Services& its derivatives -
 Veterinary Services
Veterinary Services -
 Our Global Dream
Our Global Dream
HOLDING COMPANY FOR BIoLOGICAL PRODUCTS & VACCINES SAE
is the only producer of vaccines & sera in Egypt and is one of the main blood banks.It is the oldest manufacturer of vaccines in Africa and the Middle East.
The name VACSERA reflects our commitment to serve the preventive medicine branch through production of vaccines and sera.

51 Wezaret EL Zeraa Street.
Agouza , Giza.
( +2 ) - 36711111
vacserateam@vacsera.com
( +2 ) - 33369872